Collagen, a large and abundant protein in the human body, is ubiquitous in various tissues, including skin, bones, muscles, blood vessels, internal organs, and cartilage. It serves as a crucial component of the human body and can be considered its life support.
Up to 28 types of collagen have been identified, with types I, II, and III accounting for over 90% of the total collagen content. Skin collagen is primarily composed of types I and III.
Collagen possesses a right-handed triple helix structure, where each peptide chain is arranged in a left-handed manner. Three left-handed α chains intertwine to form the right-handed helix of collagen, resulting in its characteristic triple helix conformation. These helices rotate around each other in a spring-like manner, forming a network. This unique structure gives collagen both strong elasticity and solid support, demonstrating how a protein's structure determines its function.
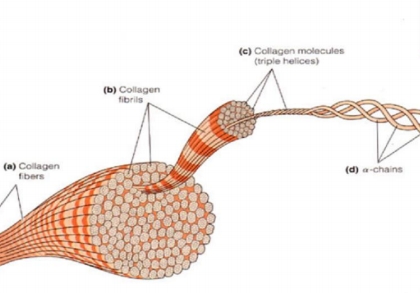
The Collagen Triple Helix Structure
The skin's collagen content peaks between the ages of 20 and 30. Following this peak, the collagen begins to degrade, declining at a rate of 1% to 1.5% per year. The concentration of type III collagen, on the other hand, starts to diminish from birth.
The Significance of Type III Collagen
|
Type I Collagen
|
Type III Collagen
|
|
crude fiber
|
fine fiber
|
|
In the shape of strips
|
Reticular
|
|
Body that nourishes the skin
|
Spread around type I collagen
|
|
Wound healing in adults is prone to scar formation
|
Baby skin heals without scars
|
|
Relatively high content, hard skin
|
Relatively high content, skin elasticity
|
|
|
|
The higher the proportion of type III collagen, the more reticulated the tissue becomes, resulting in enhanced elasticity and a firmer skin appearance.
Sources of Collagen
Plant-Based Collagen: It's worth noting that all collagen originates from animals. Plants do not possess such a tissue or protein. The term "plant collagen" typically refers to carbohydrate proteins derived from plants like peach gum and white fungus, which are not true collagen. Additionally, recombinant technologies utilizing plants, such as tobacco, as host cells to produce animal collagen, are not encompassed in this classification.
Animal-Based Collagen: The vast majority (99%) of commercially available collagen is derived from animal tissues, including pig skin, cowhide, donkey skin, fish skin, and fish scales. These animal-based collagens exhibit fundamental differences in their gene sequences compared to human collagen, classifying them as non-homologous substances. These differences pose potential risks of rejection, sensitization, and viral transmission.
Recombinant Collagen-Like Protein: Obtained through genetic recombination and bioengineering techniques, collagen-like proteins closely resemble human collagen in terms of structure and characteristics. However, due to modifications in its structure, it may be challenging to retain all of its efficacy, and there is a certain risk of sensitization.
Recombinant Humanized Collagen: This type of collagen possesses a gene sequence that aligns with human collagen, ensuring excellent tissue compatibility. It is recognized by the human body's immune system as its own substance, allowing it to smoothly penetrate the skin barrier and be directly absorbed. It participates in constructing collagen at the muscle's foundation, boasting high utilization and robust activity.
Recombinant Type III Humanized Collagen
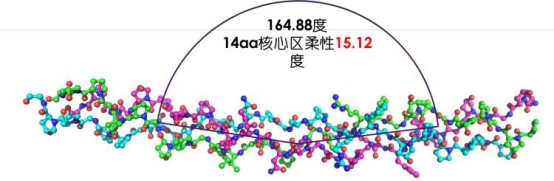
Structure
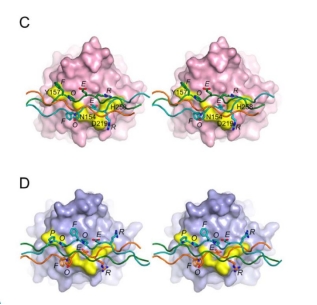
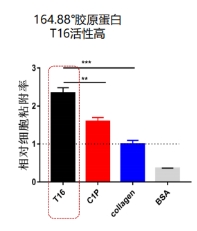
Recombinant Type III Humanized Collagen exhibits a functional region comprising a 164.88° flexible curved triple helix structure.
Sequence
The repeating unit closely aligns with the amino acid sequence of human collagen, ensuring a high level of purity.
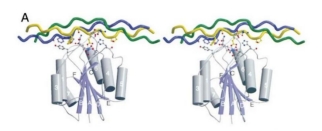
Integrin Binding
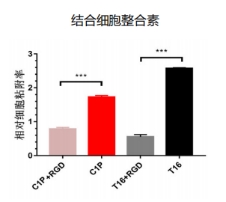
This collagen boasts numerous integrin recognition sites, including GPAGEK, GAPGER, and GPAGFR, allowing for enhanced interaction with fibroblast receptors. This strengthened integrin binding further regulates cell recognition, adhesion, and migration. The flexible curvature of this humanized collagen favors superior binding with cell receptor proteins.
Cellular Adhesion
Cells adhere firmly to Recombinant Type III Humanized Collagen, demonstrating its high biological activity.
High Activity
The collagen retains robust biological activity, making it a highly effective choice.
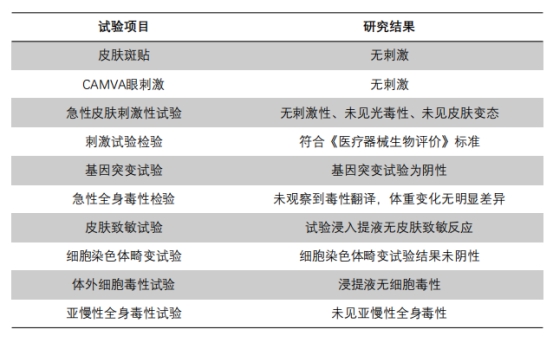
Security Research
Functions
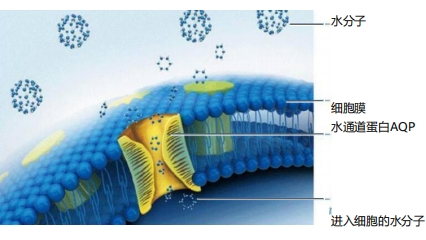
1. Moisturizing and Hydrating Properties
Research indicates that the AQP-2 protein regulates water transport across cell membranes, earning it the moniker of aquaporin 2. Our highly active recombinant type III humanized collagen enhances the expression of AQP-2, beneficial to damaged cellular water channels and further promoting hyaluronic acid synthesis.
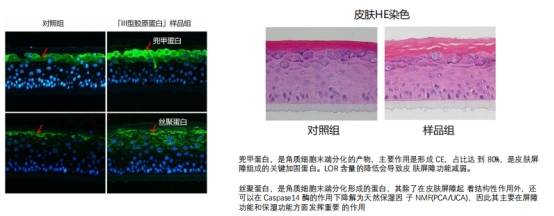
2. Enhancing Skin Improvement
This collagen significantly thickens the stratum corneum, bolstering skin barrier function and upregulating the expression of loricrin and filaggrin.
3. Suppressing Decline
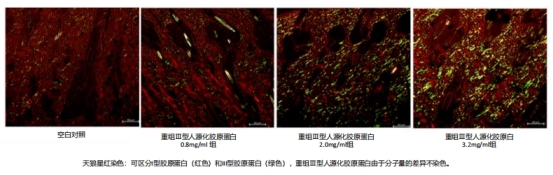
Studies reveal that upon integration with the extracellular matrix (ECM) for 8 weeks, recombinant type III humanized collagen significantly stimulates the regeneration of type I and III collagen in tissues. The regenerative effect varies with the concentration administered.
Literature Insights
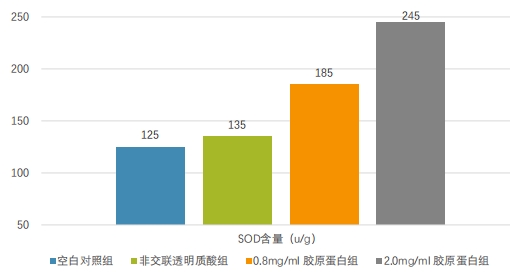
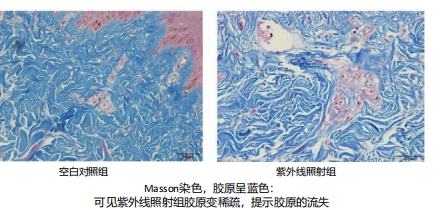
In the rat skin ultraviolet photoaging model, ultraviolet irradiation triggers the production of ROS (reactive oxygen species) and diminishes SOD (superoxide dismutase) activity, ultimately causing DNA damage in skin tissue. However, the introduction of recombinant type III humanized collagen has been shown to mitigate photoaging effects and boost SOD activity.
Furthermore, research indicates that this humanized collagen decreases the levels of MMPs (matrix metalloproteinases), effectively reducing collagen degradation.
Application of recombinant type III humanized collagen
skin improvement
Improvement after sun exposure
oral improvement
Medical beauty
Eye cream, etc.