About Seebio
Pioneer of Core Raw Materials for Functional Food Ingredients and Healthcare Supplements in China.
|
Item Number
|
Product
|
λmax(nm)
|
Molar absorbance
|
|
DCD0049A
|
N-(2-Hydroxy-3-sulfopropyl)-3,5-dimethoxyaniline sodium salt; HDAOS, 82692-88-4
|
583
|
1.73x104
|
|
DCD0006A
|
3-(N-Ethyl-3-methylanilino)-2-hydroxypropanesulonic acid sodium salt; TOOS, 82692-93-1
|
555
|
3.92x104
|
|
DCE0552A
|
N,N-Bis(4-sulfobutyl)-3,5-dimethylaniline disodium salt; MADB, 209518-16-1
|
630
|
1.65x104
|
|
ACC0083A
|
N-Ethyl-N-(2-hydroxy-3-sulfopropyl)-3,5-dimethylaniline sodium salt monohydrate; MAOS, 82692-97-5
|
630
|
2.25x104
|
|
DCC0040A
|
N-Ethyl-N-(3-sulfopropyl)-3-methoxyaniline sodium salt; ADPS, 82611-88-9
|
540
|
2.79x104
|
|
ACD0074B
|
N,N-Bis(4-sulfobutyl)-m-toluidine, disodium salt; TODB, 1044537-70-3
|
550
|
3.80x104
|
|
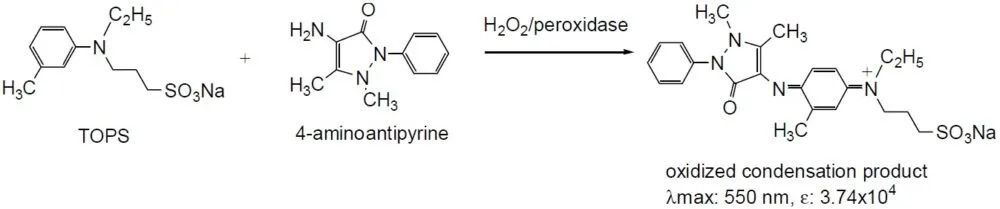
DCD0035A
|
N-Ethyl-N-(3-sulfopropyl)-3-Methylaniline Sodium Salt; TOPS, 40567-80-4
|
550
|
3.74x104
|
|
DCD0049A
|
N-Ethyl-N-(2-hydroxy-3-sulfopropyl)-3 5-dimethoxyaniline sodium salt; DAOS, 83777-30-4
|
593
|
1.75x104
|
|
DCD0004A
|
Nitro Blue Tetrazolium; NBT, 298-83-9
|
530
|
3.6x104
|
|
DFG0001A
|
Iodonitrotetrazolium chloride; INT, 146-68-9
|
490
|
1.5x104
|
|
ACD0021A
|
Blue tetrazolium chloride; BT, 1871-22-3
|
525
|
2.6x104
|